Experience can modify connectivity patterns, resulting in modifications of behaviour. This enables memory as we learn and adaptation to our changing environments as we go through life. Conversely, in brain diseases these processes can be impaired and overridden by synaptic, neurite and cell loss. Thus, physical, material changes in cells modify how the brain works. We want to understand how the brain changes throughout life, the underlying molecular mechanisms and how this modifies behaviour.
A Toll receptor map underlies structural brain plasticity
Structural brain plasticity is encoded in the topographic distribution of Toll receptors and their ability to switch between alternative signalling outcomes, thus translating diverse sensory experience into structural change.
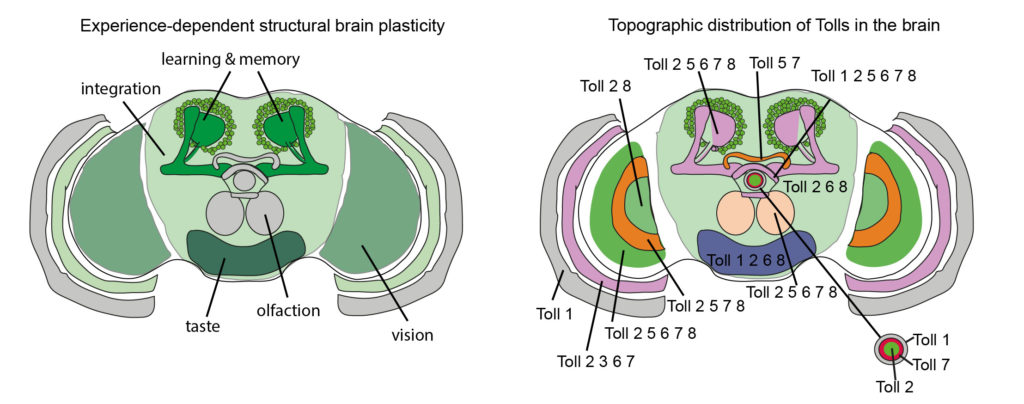
Structural plasticity reveals that brain function is encoded in generative changes to cells. Others had previously shown that at an adult critical period, experience increases fiber number and brain size in Drosophila. We asked if Toll receptors are involved. We found that most Tolls are expressed in the adult brain, where they demarcate a map of brain anatomical domains. Toll-2 gain of function and neuronal activity at the critical period increased cell number and brain size. Through their topographic distribution, Toll receptors regulate neuronal number and brain size, modulating structural plasticity in the adult brain.
Tolls and neurodegeneration
Loss of function for Toll-2 during CNS development resulted in neuronal apoptosis, neuronal loss, impaired locomotion and reduced longevity – all hallmarks of neurodegeneration. Knock-down of multiple Tolls at once during development, resulted in dramatic decrease in the size of distinct brain modules. Thus, brain formation requires Toll receptor function and their deficit can cause neurodegeneration.
In the larval CNS, pro-DNTs and some Tolls can also induce apoptosis. This means that signalling via Toll receptors and their ligands can either prevent or promote neurodegeneration.
Adult neurogenesis in the Drosophila brain
Whether the brain of flies or humans can generate new neurons throughout life, has long been controversial. Finding this out is very important to understand the brain. What neural circuits do newly generated neurons in an adult integrate into? are they functional? and what is the consequence for behaviour? But it could also have tremendous impact on finding new ways to tackle brain injury and disease. Even if adult neurogenesis were not to happen, if we understood how to induce neurogenesis, we could find new ways to promote regeneration and repair. The reason this question is still controversial, is because finding convincing evidence that adult neurogenesis takes place is extremely difficult. We demonstrated that knock-down of Toll receptors during brain development resulted in neurodegeneration and dramatic reduction in brain size. We provided evidence that when Toll-2 and its non-immune downstream signalling pathway is activated only in the adult, neurogenesis could be induced at a critical period in the adult brain, that this involved cell proliferation and it resulted in increased brain size. Our data indicate that even if adult neurogenesis were to be present although scarce in the adult brain, Toll receptor signalling can induce it.
How does experience shape the brain?
We are now busy trying to answer this question and the underlying molecular mechanisms. Watch this space!
To find out more, read:
Li G, Forero MG, Wentzell JS, Durmus I, Wolf R, Anthoney NC, Parker M, Jiang R, Hasenauer J, Strausfeld NJ,Heisenberg M, Hidalgo A (2020) A Toll-receptor map underlies structural brain plasticity eLife, 9: e52743 DOI: 10.7554/eLife.52743
- eLife Digest article 17 March 2020 dedicated to our paper: “How experience shapes the brain”
https://elifesciences.org/digests/52743/how-experience-shapes-the-brain
Li G and Hidalgo A (2020) Adult neurogenesis in the Drosophila brain: the evidence and the void. International Journal of Molecular Sciences 21(18), 6653
Guiyi Li and Alicia Hidalgo (2021) The Toll route to structural brain plasticity. Frontiers in Physiology. Frontiers in Physiology DOI: 10.3389/fphys.2021.679766
Funded by:
BBSRC
Marie Curie-Sklodowska Fellowship (Jun Sun).
Wellcome Trust Investigator Award (Alicia Hidalgo)